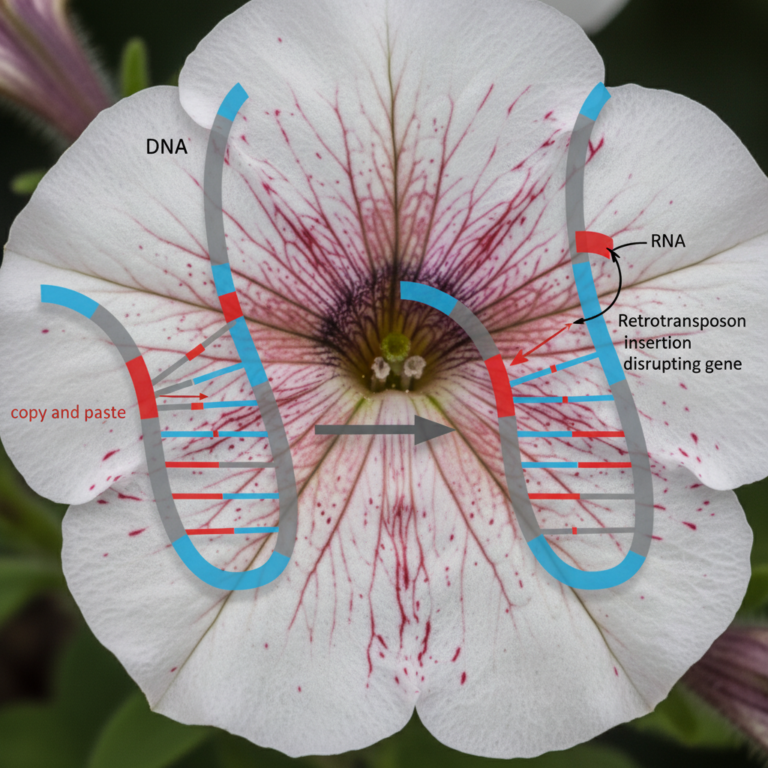
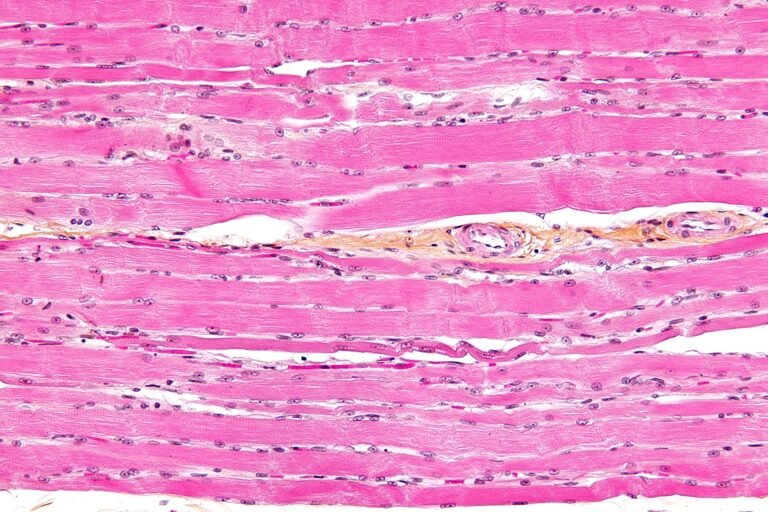
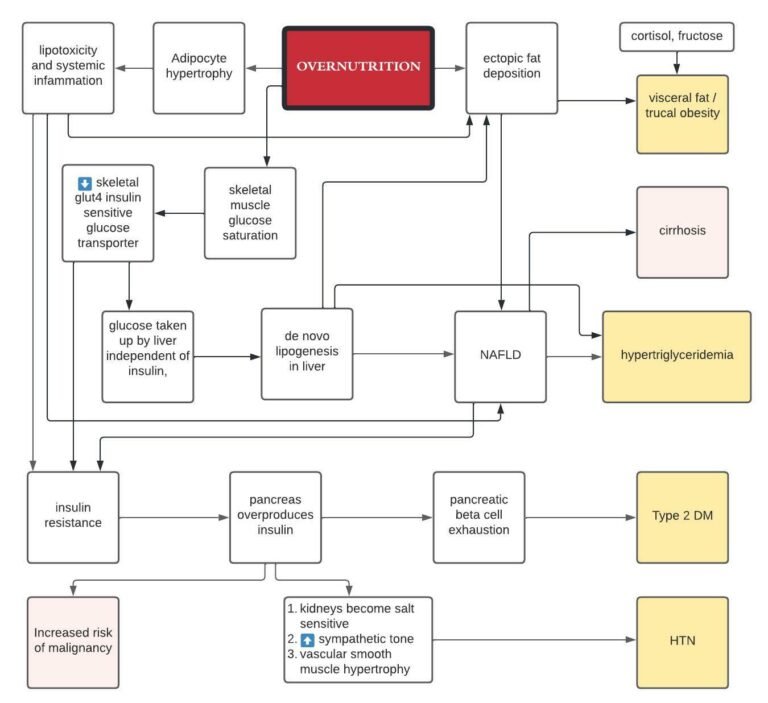
The Question of Protein
I am everywhere surrounded by friends and family sprinkling creatine onto their protein shakes and shoveling it into their mouths as they compulsively pump iron in an endless attempt to build muscle. The societal obsession with protein and muscle growth can be directly traced to Peter Attia, who in turn was influenced by a man…