Not a week goes by that I don’t get a question or an IG post or some Huberman / Rogansphere related podcast link about supplementing NAD. Often from the same individual actually. Is it worth spending your money to take NMN or NR or niacin or any one of the many varieties of this cofactor?
What NAD+ is and actually does
- Cellular metabolism: NAD+ is a coenzyme, meaning it works alongside enzymes rather than acting on its own. It exists in two forms—oxidized (NAD+) and reduced (NADH)—and the cell uses the conversion between them to shuttle electrons. Glycolysis, the citric acid cycle, beta-oxidation of fatty acids: all of them generate ATP only because NAD+ is there to accept electrons and ferry them to the electron transport chain. Without NAD+, mitochondria stop and you don’t make energy. But most people have enough NAD+. What is more important is the ratio of NAD+ to NADH.
In a healthy cytoplasm, the free NAD+/NADH ratio is enormous, somewhere on the order of 700:1. In the mitochondrial matrix it is much lower, around 7:1 to 10:1. If you have lots of NAD+ relative to NADH, oxidativereactions proceed—glucose gets oxidized, fatty acids get burned, electrons flow into the mitochondrial respiratory chain. If you have lots of NADH relative to NAD+, the cell is signaled to stop oxidizing and start storing—fatty acid synthesis, gluconeogenesis from lactate, building rather than breaking down.
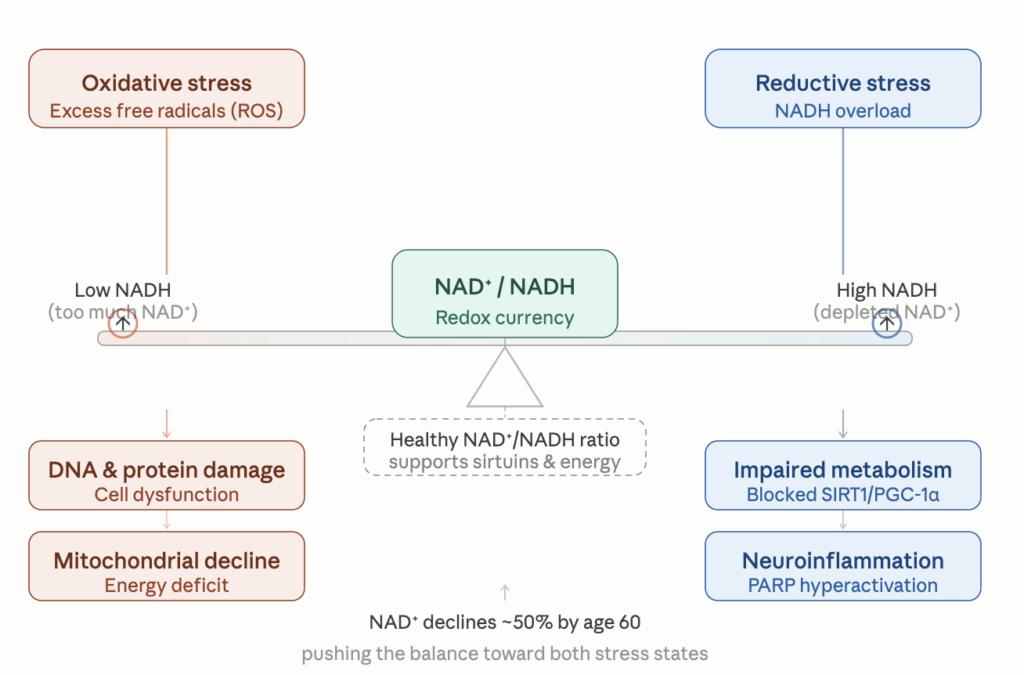
Most discussions of metabolic damage focus on oxidative stress—the accumulation of reactive oxygen species. Less appreciated, but possibly equally important in modern disease, is its mirror image: reductive stress, the state of having too much NADH and not enough NAD+.
Chronic overfeeding produces exactly this. When you continuously supply substrate—glucose, fatty acids, amino acids—to a cell that is not actually using much energy, glycolysis and beta-oxidation generate NADH faster than the electron transport chain can dispose of it. The NAD+/NADH ratio collapses. Several things happen as a consequence: The mitochondria back up. Electron transport slows because complex I cannot offload electrons from NADH fast enough, and the ones that do get loaded tend to leak out as superoxide—the actual source of much of the reactive oxygen species we worry about. Paradoxically, reductive stress causes oxidative stress.
- DNA Repair: it is believed by some and not by others that the NAD/NADH ratio affects the function of PARPs which are all DNA repair enzymes. The PARPs (poly-ADP-ribose polymerases) detect DNA damage and recruit repair machinery. Every time a strand break occurs—and in a typical cell this is thousands of times a day—PARPs consume NAD+ to flag the damage. Less NAD means less DNA repair, which means faster aging.
- The sirtuins (SIRT1 through SIRT7) are deacetylases that remove acetyl groups from histones and other proteins. They are central to the cellular response to caloric restriction, they tune mitochondrial biogenesis, and they regulate the expression of stress-response genes. Sirtuins cannot function without NAD+.
- Inflammation: CD38, an ectoenzyme present on immune cells and many other tissues, also consumes NAD+. CD38 expression rises substantially with age and with chronic inflammation, and it is now thought to be one of the principal reasons that NAD+ levels fall as we get older.
So NAD+ is being burned through multiple different ways at once—to make energy, to repair DNA, and to fuel an inflammatory enzyme that gets busier with age—while the cell’s capacity to synthesize it appears to decline. Tissue measurements suggest NAD+ levels in skin, muscle, liver, and brain fall by something on the order of 50% between young adulthood and old age. The hypothesis that follows is straightforward: if we can refill the tank, perhaps some of the downstream consequences of low NAD+—mitochondrial dysfunction, impaired DNA repair, chronic inflammation—can be reversed or slowed.
The four oral options
You cannot simply swallow NAD+ and expect it to reach your cells. The molecule is too large and too charged to cross membranes intact; it gets broken down in the gut. So all oral strategies rely on precursors that the body can transport and assemble into NAD+ on the inside.
- Niacin (nicotinic acid) is the original B3 vitamin, the one that cures pellagra. It enters the Preiss-Handler pathway and is reliably converted to NAD+ in the liver and elsewhere. It is cheap, well-studied, and has the inconvenient property of producing a vasodilatory flush—the prostaglandin-mediated burning, itching sensation that has caused generations of patients to abandon it.
- Niacinamide (nicotinamide, NAM) is the amide form of niacin. It does not cause flushing because it does not act on the same receptor (GPR109A) that mediates the flush. It is converted to NAD+ via the salvage pathway. The catch: at high doses, niacinamide actually inhibits sirtuins. It is a sirtuin product, and product inhibition is exactly what you would expect. So while niacinamide raises NAD+, it may blunt one of the downstream pathways you are trying to activate.
- Nicotinamide riboside (NR) is a more recently characterized precursor that bypasses some of the rate-limiting steps in the salvage pathway. Multiple human trials have shown that 300–1000 mg per day of NR reliably raises whole-blood NAD+ levels, is well tolerated, and produces modest improvements in markers like blood pressure and arterial stiffness in older adults.
- Nicotinamide mononucleotide (NMN) is one step further down the synthesis pathway than NR. The question of whether NMN itself crosses cell membranes or whether it must first be converted back to NR has been debated for years; the practical answer seems to be that oral NMN does raise NAD+ in humans. A randomized trial in middle-aged adults given 250 mg per day showed elevation of blood NAD+ and prevention of an age-related rise in HOMA-IR (an insulin resistance score) seen in the placebo group. A study in older Japanese men using 250 mg per day for twelve weeks showed improvements in muscle function on certain measures.
The honest summary of the oral precursor literature is this: all of them raise NAD+ in blood, the safety profiles look good across studies running up to a year, and the clinical effects on hard outcomes—cardiovascular events, cognitive decline, mortality—have not yet been demonstrated in adequately powered trials. The studies showing improvements tend to be small, of short duration, and heterogeneous in their endpoints. We are still in the phase where we can say with confidence that these compounds do what they say on the tin biochemically, but not yet that they translate into the kind of healthspan extension that the mouse data suggest is possible.
The IV NAD+ DANGER
A growing cottage industry offers intravenous NAD+ at concierge clinics and longevity spas, often at considerable expense. The pitch is appealing—why mess around with precursors when you can deliver the finished molecule directly?
The instinct that something is off about putting a lot of NAD+ into the bloodstream turns out to be biochemically grounded. Inside the cell, NAD+ is one of the most abundant small molecules in metabolism. Outside the cell, it is something else entirely: a damage-associated molecular pattern, or DAMP. When cells lyse from infection, trauma, or necrosis, they spill their NAD+ into the extracellular space, and the immune system reads this as a signal that something has gone wrong. Extracellular NAD+ triggers inflammatory and immune responses.
This explains the well-known side effect profile of IV NAD+: nausea, malaise, abdominal cramping, sweating, anxiety, chest pressure. These are not idiosyncratic reactions—they are predictable consequences of pushing extracellular NAD+ to supraphysiologic concentrations and activating purinergic signaling. The standard clinical workaround is to infuse very slowly, often over four to six hours, which keeps extracellular concentrations from spiking high enough to trigger the worst of it.
A recent retrospective comparison of IV NAD+ versus IV NR in a real-world clinic setting found that NR could be delivered considerably faster with fewer adverse experiences, and neither produced clinically significant changes in liver enzymes or inflammatory markers at 30 days. This is reassuring as far as it goes, but the studies remain small, short, and uncontrolled. There are no randomized controlled trials demonstrating that IV NAD+ produces clinical benefits beyond what oral precursors provide, and the cost differential is enormous. My own view is that the burden of proof for IV NAD+ is on the people charging $800 a session for it, and that burden has not been met.
The most provocative piece of clinical evidence in this entire field comes not from a longevity study but from a 1960s cholesterol trial. The Coronary Drug Project enrolled 8,341 men with prior myocardial infarction and randomized them to placebo or one of several lipid-lowering interventions, including 3 grams per day of niacin. The trial ran from 1966 to 1975. At the end of the active treatment period, niacin showed a modest reduction in nonfatal MI but no significant mortality benefit. A reasonable result. The trial closed.
Then in 1985, nine years after everyone had stopped taking the drug, the investigators did a follow-up to determine vital status on the original cohort. What they found was unexpected enough that it has been discussed ever since. All-cause mortality in the niacin group was 11% lower than in the placebo group (52.0% vs 58.2%, p=0.0004). The benefit was present across every major category of death—coronary, other cardiovascular, cancer, and other. Translated into life expectancy, the niacin recipients had gained roughly 1.6 years of additional life. And remember: they had stopped taking the drug almost a decade earlier.
A late, durable, all-cause mortality benefit from a drug discontinued years before is a strange finding. The conventional explanation has been that niacin’s lipid effects produced a slow accrual of cardiovascular benefit. But the cancer mortality reduction and the cross-category nature of the effect have always sat awkwardly with a pure lipid-lowering story. Looked at through an NAD+ lens, the result is more interpretable: a sustained pharmacologic boost of NAD+ during a critical window may have produced cumulative benefits—better DNA repair capacity, better mitochondrial maintenance, lower CD38-driven inflammation—that compounded over the subsequent decade. This is speculative. It is not what the trial was designed to test. But it is the kind of result that makes you take the pathway seriously.
It is worth noting that subsequent niacin trials in the statin era—AIM-HIGH and HPS2-THRIVE—failed to show cardiovascular benefit when niacin was added on top of statin therapy. Whether this reflects a true loss of benefit in the modern lipid-lowered population, or simply the use of formulations and dosing that did not replicate the original CDP exposure, is debated. The CDP result remains the single most striking long-term outcomes signal in the entire NAD+ literature, and it is over forty years old.
Where this leaves us
Cellular NAD+ falls with age. Restoring it in mice produces effects that look a lot like rejuvenation across multiple tissues. In humans, oral precursors reliably raise cellular NAD+ levels, appear safe over the durations studied, and produce small signals in surrogate markers—blood pressure, arterial stiffness, insulin sensitivity, certain measures of muscle function. They have not yet been shown to extend healthspan or lifespan in adequately powered trials, and such trials are difficult to run at the scale and duration required.
For a patient asking what to do today, my reading is roughly this: the case for some form of B3 supplementation in older adults is reasonable, given the favorable safety profile and the mechanistic plausibility. NR and NMN are the most studied of the modern precursors. Niacin remains the only B3 compound with long-term hard outcomes data, and that data is genuinely impressive even if it is decades old; the flush is the price of admission. High-dose niacinamide is probably best avoided as a longevity strategy given its sirtuin inhibition. IV NAD+ is, in my opinion, at best a way to spend money. At worst, you are doing physical harm to your body.
For young people who are not depleted in NAD, supplementation seems superfluous. If the ratio is what matters, what should we expect from oral precursors? They certainly raise the total NAD+ pool. But unless you also do something to keep NADH from accumulating, much of the new NAD+ may simply be reduced to NADH by the substrate the cell is already drowning in. You’ll have a bigger pool, with the same unfavorable distribution. Concentrate instead on raising the NAD / NADH level through exercise and periodic fasting.
