Advanced Glycation End Products
I have occasionally complained about my mother in law’s bland cooking methods. Boiled this, steamed that. How about a sauté? How about frying something? And yet, she is aging gracefully and the scientific evidence has begun to justify her insipid cooking.
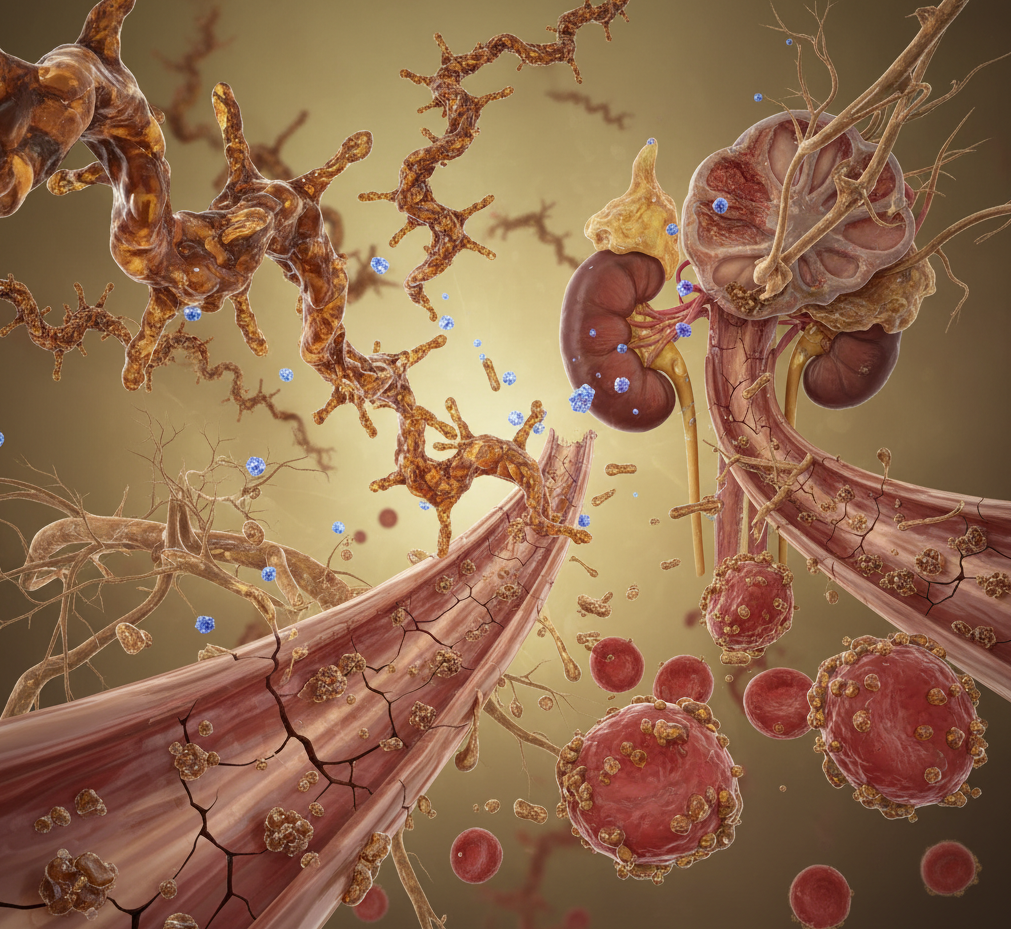
A growing body of research shows that advanced glycation end products (AGEs) sit at the crossroads of metabolic dysfunction, chronic inflammation, and accelerated aging. They form when sugars bind to proteins or lipids through non‑enzymatic reactions, creating irreversible compounds that accumulate in tissues over time. High‑temperature cooking, processed foods, hyperglycemia, and oxidative stress all accelerate their formation.
How AGEs Form and Why They Accumulate
AGEs arise from the Maillard reaction—initially a browning reaction in foods, but also a slow, ongoing process inside the body. They come from two sources:
- Exogenous AGEs
from foods cooked at high temperatures (grilling, frying, roasting) and ultra‑processed products. - Endogenous AGEs
generated in vivo, especially when glucose levels run high or oxidative stress is elevated. Oxford Academic
In humans, endogenous AGE’s predominate. Exogenous AGEs from food matter too, but their impact is smaller and more modifiable.
Why AGEs Matter for Metabolic Health
AGEs are not passive byproducts; they actively disrupt metabolic homeostasis through several mechanisms:
- Chronic Inflammation via RAGE Activation
AGEs bind to the receptor for advanced glycation end products (RAGE), triggering inflammatory signaling cascades. This amplifies oxidative stress and promotes a persistent low‑grade inflammatory state—one of the hallmarks of metabolic syndrome.
- Impaired Insulin Sensitivity
By damaging insulin receptors and altering intracellular signaling, AGEs contribute to insulin resistance. High AGE levels are strongly linked to diabetes and its complications.
- Structural Damage to Metabolic Tissues
AGEs crosslink collagen and other structural proteins, stiffening blood vessels, impairing microcirculation, and disrupting extracellular matrix integrity. This affects adipose tissue remodeling, pancreatic β‑cell function, and vascular health.
- Mitochondrial Stress and Reduced Metabolic Flexibility
Oxidative stress induced by AGEs impairs mitochondrial function, reducing the body’s ability to switch efficiently between fuel sources—an early feature of metabolic decline.
- Acceleration of Age‑Related Metabolic Diseases
AGE accumulation is associated with diabetes, cardiovascular disease, kidney dysfunction, and neurodegeneration—conditions that share overlapping metabolic pathways.
The Dietary Connection: How Food Choices Influence AGE Load
Cooking methods dramatically influence AGE content:
- High‑AGE methods:
frying, broiling, grilling, roasting - Low‑AGE methods:
steaming, boiling, poaching, slow‑cooking
Processed foods rich in sugars and fats are particularly dense in AGEs. Reducing dietary AGE intake has been shown to improve markers of inflammation and metabolic health.
Strategies to Reduce AGE Burden
Research highlights several interventions that meaningfully lower AGE accumulation:
- Lower‑temperature cooking
and moisture‑rich methods - Higher intake of antioxidants
(fruits, vegetables, herbs) to counter oxidative stress - Increased dietary fiber
, which improves glycemic control and reduces endogenous AGE formation - Regular physical activity
, which improves glucose handling and reduces oxidative load - Limiting ultra‑processed foods
and added sugars
Emerging therapies aim to inhibit AGE formation or block RAGE signaling, but lifestyle strategies remain the most accessible and evidence‑supported tools.
Glylo is one evidence based option to reduce the burden of AGE’s that’s commercially available.
The AgeProof Takeaway
AGEs are not just biochemical curiosities—they are active drivers of metabolic dysfunction and accelerators of biological aging. They damage tissues, amplify inflammation, impair insulin signaling, and undermine mitochondrial resilience. The good news: AGE burden is modifiable. Small shifts in cooking methods, food choices, and metabolic health behaviors can meaningfully reduce exposure and slow the metabolic wear‑and‑tear that AGEs impose.
What is the effect size of AGE’s?

 The two popular theories that purport to explain the obesity pandemic are the carbohydrate insulin model and the standard model of energy balance. In Why Nature Wants Us to be Fat, nephrologist Richard Johnson bravely proposes another theory: the survival switch.
The two popular theories that purport to explain the obesity pandemic are the carbohydrate insulin model and the standard model of energy balance. In Why Nature Wants Us to be Fat, nephrologist Richard Johnson bravely proposes another theory: the survival switch.